
By Dr Martine Stoffels, consultant neuropsychiatrist and clinical director, Phoenix Mental Health Services
Repetitive transcranial magnetic stimulation (rTMS) has travelled an extraordinary path, from a niche neuroscience tool to a frontline therapy in treatment-resistant depression.
But could it now reshape the future of neurorehabilitation?
Dr Martine Stoffels, Consultant Neuropsychiatrist and Clinical Director at Phoenix Mental Health Services UK, argues that rTMS is on the cusp of becoming a core element of brain-injury rehabilitation, provided it is applied within a structured, consultant-led framework.
She says: “We’re seeing mounting evidence that rTMS can modulate brain networks disrupted after injury.
“Not just improve mood, but also enhance attention, executive function, and drive neuroplastic recovery.
“The key is delivering it as part of a properly governed neurorehabilitation model, not as a standalone gadget.”
The Unmet Need in Brain Injury Rehabilitation
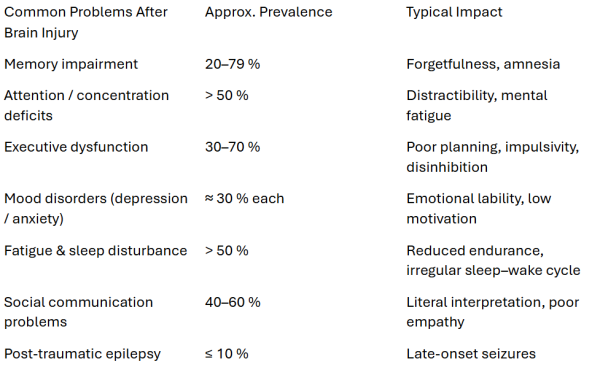
Every year, thousands of people in the UK live with the long-term neuropsychiatric fallout of acquired brain injury (ABI).
Mood instability, apathy, fatigue, impulsivity, and cognitive inefficiency often persist long after physical recovery.
Traditional rehabilitation, while effective for functional outcomes, remains limited in restoring cognitive-emotional integration.
rTMS uses electromagnetic induction to deliver focused pulses to cortical neurons, modulating activity within specific brain networks.
Depending on frequency and target, it can increase or decrease cortical excitability, effectively “retuning” circuits disrupted by injury.
By doing so, rTMS supports neuroplasticity – the brain’s intrinsic ability to reorganise and form new connections after injury.
Evidence Base: From Depression to Neurorehab

rTMS in Brain Injury: Clinical Application
In neurorehabilitation, the goal is not just symptom relief but functional reintegration —rebuilding independence, emotional control, and cognitive stamina.
Dr Stoffels emphasises that rTMS should be delivered within a consultant-supervised framework, where stimulation parameters are tailored to the neuropsychiatric profile and integrated with ongoing therapy.
Seizure Risk and Safety
Given the prevalence of post-traumatic epilepsy, safety is a legitimate concern.
Large-scale reviews report rTMS-induced seizures in < 0.1 % of sessions – comparable to antidepressant trials.
Governance and consultant oversight ensure safety, particularly in complex neurological populations.
Beyond Depression: Treating the Whole Brain
Patients frequently report clearer thinking, less cognitive fog, and greater emotional stability, outcomes not easily achieved through medication alone.
Functional MRI studies corroborate these findings, showing enhanced prefrontal activity and improved synchrony with parietal and limbic networks following stimulation.
Case Illustration: Bringing Neuromodulation Home
At Phoenix Mental Health Services, rTMS is no longer confined to clinic walls.
One striking case involved a quadriplegic patient with a severe head injury, who presented with treatment-resistant depression and marked attentional deficits.
Traditional pharmacological approaches had provided minimal benefit, and profound mobility limitations made attending clinic-based sessions impossible.
Through Phoenix’s consultant-led community programme, a portable rTMS unit was delivered and monitored within the patient’s home.
Over a structured six-week course, with stimulation targeted to the left dorsolateral prefrontal cortex, the patient demonstrated clear improvements in mood, attention, and concentration, as well as a notable reduction in spasticity observed by physiotherapists and carers.
These gains translated into better engagement with daily routines, improved emotional regulation, and an overall lift in communication and social participation.
Home-delivered rTMS exemplifies how consultant-supervised private practice can bridge the gap between hospital-level expertise and community-based recovery.
Barriers to Implementation
Despite a steadily expanding evidence base, rTMS remains underutilised in UK neurorehabilitation.
Several interlinked factors appear to limit its wider adoption.
First, professional familiarity remains low. Most consultants working in neuropsychiatry and neurorehabilitation receive little formal training in neuromodulation, and rTMS continues to be perceived by some as an emerging or experimental technique rather than an established clinical intervention.
This knowledge gap contributes to ongoing uncertainty about its indications, optimal protocols, and safety in complex neurological populations.
Second, practical barriers constrain service delivery. Standard protocols typically require 10–30 sessions of around 30 minutes each, often delivered daily.
For individuals with significant fatigue, mobility restrictions, or cognitive impairments, the logistics of attending frequent sessions can be prohibitive.
Only a small number of UK services currently possess the necessary equipment and consultant expertise to deliver rTMS in either clinic or community settings.
Third, commissioning and funding processes present significant challenges. Decisions are often made by case managers or professional deputies, who may have limited awareness of the clinical rationale or evidence underpinning rTMS.
As a result, proposals can become the subject of extended negotiation between legal teams and insurers, delaying treatment access.
In medico-legal contexts, such delays are compounded by the time required to instruct a consultant with both neuropsychiatric and neuromodulation expertise, which may take several
months.
Timing is crucial. Evidence suggests that neuromodulatory interventions are most effective when initiated within weeks to months of the initial injury, during the period of heightened
neuroplasticity.
In practice, however, many patients are only considered for rTMS years after injury, by which point potential gains may be diminished.
Finally, logistical and governance limitations, including requirements for equipment calibration, technician training, and ongoing consultant supervision, restrict scalability.
Community delivery, though feasible, depends on portable devices and robust clinical oversight to maintain safety and efficacy standards.
Together, these factors illustrate that the barriers to implementation are systemic rather than scientific: the evidence supports rTMS, but infrastructure, awareness, and commissioning
frameworks have yet to catch up.
Future Developments
The next frontier lies in precision neuromodulation – combining rTMS with EEG-guided mapping, real-time connectivity analysis, and AI-driven personalisation.
Trials are exploring theta-burst stimulation (TBS), a shorter, high-intensity variant offering comparable efficacy in a fraction of the time. Integration with pharmacogenetic testing, nutritional neuroscience, and wearable monitoring may further individualise treatment.
Within five years, rTMS could become a mainstay of post-acute neurorehabilitation, provided governance keeps pace with innovation.
References
Fitzgerald P B et al. (2022). The efficacy of rTMS in post-traumatic depression and executive dysfunction. J Neuropsychiatry Clin Neurosci.
Kang N et al. (2020). Cognitive enhancement through rTMS in TBI survivors. Front Neurol.
Lee W H et al. (2022). rTMS in Alzheimer’s and vascular dementia: meta-analytic review. Brain Stimul.
Leung J et al. (2023). High-frequency DLPFC stimulation for post-TBI depression. Neurorehabil Neural Repair.
Manganotti P et al. (2021). Neuromodulation for apathy and fatigue syndromes. Restor Neurol Neurosci.
Sabbagh M et al. (2020). Network modulation and cognition in mild Alzheimer’s disease. Cortex.
Author Bio
Dr Martine Stoffels is a Consultant Neuropsychiatrist and Clinical Director at Phoenix Mental Health Services UK.
Her clinical interests include acquired brain injury, neuropsychiatric rehabilitation, neuromodulation, and the integration of psychiatry with neuropsychology and occupational therapy.
She is currently developing Velaris Integrated Interventions, a consultant-led model for rapid, multidisciplinary neurorehabilitation across the UK.








