Functional Electrical Stimulation (FES) has transformed the lives of thousands of UK patients who’ve been able to access it.
However, it remains out of reach for some NHS patients. Odstock Medical Limited (OML) is on a mission to change that.
The company has been at the forefront of FES innovation since 2005.
Its technology was born out of Salisbury NHS Foundation Trust research, led by OML co-founder and medical director, Dr Paul Taylor.
Paul says: “We developed FES in the NHS for the NHS.
“But although FES is in the national guidelines, it isn’t universally funded in the NHS.”
It’s a frustrating situation, both for OML and the potential thousands of patients who could benefit from the technology.
But the situation has galvanised Paul and his team to keep refining their devices and producing evidence of their safety and efficacy.
How does it work?
FES works by stimulating the nerves that control paralysed or weakened muscles using external electrodes, similar in appearance to the sticky pads used in TENS machines.
The stimulation is precisely timed to support movement, most often to assist with walking or hand function.
FES can be used for a wide range of conditions, including stroke, MS, spinal cord injury, Parkinson’s and cerebral palsy. It can be used for both adults and children.
Ultimately, it may benefit almost any neurological condition that disrupts the flow of signals from the brain to the body.

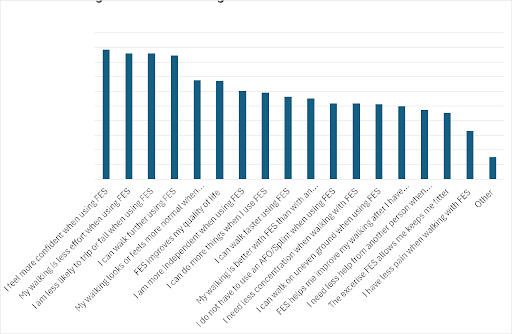
Patient survey 2025 results
OML conduct an annual FES user survey to record opinions regarding the service, the equipment and the effects of the treatment. A questionnaire was sent to a random selection of 398 service users. OML received 161 responses.
Key highlights:
- 84 per cent of FES users report a reduction in the incidence of falls after using FES.
- The most frequently cited reason for using FES was improved confidence (88 per cent) while walking. This is likely associated with an increased sense of safety during walking, linked to a reduced risk of falls (86 per cent) and decreased walking effort (86 per cent), which were jointly the second most commonly cited reasons, alongside increased walking distance (85 pr cent).
STEPS II Parkinson’s trial
OML is spearheading an ongoing multi-site clinical study exploring whether FES can improve walking in people with Parkinson’s disease.
STEPS II builds on earlier, preliminary research which suggested that the technology helped patients walk faster and reduced symptoms, even having an impact the next day in some cases.
STEPS II is a collaboration with eight centres across the UK, coordinated by the Peninsula Clinical Trials Unit at the University of Plymouth.
The team aims to recruit 234 participants, with that figure now very much within reach.
Paul says: “I have no feel for the overall results of the study.
“It’ll be a genuine reveal moment for me as well as anyone else.”
Unpicking outcomes
While walking speed is an obvious outcome measure, existing FES patients often cite other benefits when explaining how the technology has helped them.
The word ‘confidence’ often comes up, Paul says, though what patients mean by that is unclear.
It seems to be something to do with the idea that walking is safer and patients feel confident that they can complete their journeys.
It’s also something to do with a reduction in effort, Paul says.
He adds: “It’s very multi-factorial, and we want to find out more about what this means, so we’ll be working with lots of researchers to unpick that.
“We’ll be looking at qualitative research techniques to really get a grip on what makes a difference to individual patients themselves.”
Looking ahead
While the results of STEPS II loom large, Paul has further ambitions for the year ahead.
OML is working on a number of projects, including one investigating the use of spinal cord stimulation in conjunction with drop foot stimulation for a longer-lasting effect.
All their endeavors, delivered in collaboration with partners around the UK, have two main aims:
For more information: