An MS trial in the US is enrolling people with relapsing-remitting MS to test a vagus nerve implant designed to reduce inflammation and promote myelin repair.
The pilot clinical trial, listed as NCT06796504, is enrolling up to 60 people aged 22 to 50 and will assess the safety and potential remyelinating effects of Setpoint Medical’s device when used alongside standard disease-modifying therapies.
David Chernoff, chief medical officer of Setpoint, said: “Our approach to activating the body’s innate neuroimmune pathways offers a compelling and novel mechanism that could complement current standards of care of multiple sclerosis.”
The first participants have been enrolled at the Shepherd Center’s Andrew C. Carlos MS Institute in Georgia and the UW Medicine Multiple Sclerosis Center in Seattle.
Recruitment is also taking place at the Minnesota Center for Multiple Sclerosis, West Virginia University, Johns Hopkins University in Maryland, UT Medicine Multiple Sclerosis and Neuroimmunology Center in Texas, and the University of Texas Health Science Center at Houston.
MS is an autoimmune condition, meaning the immune system mistakenly attacks the body.
In MS, inflammation damages myelin, the protective coating around nerve fibres in the brain and spinal cord, disrupting the signals nerves use to communicate.
Current disease-modifying therapies are intended to reduce inflammation, lower relapse risk and limit new disease activity seen on MRI scans. Many can slow the accumulation of disability over time, but they do not directly repair damaged myelin.
Jacqueline Rosenthal, medical director of the MS Institute and principal investigator of the study at Shepherd Center, said: “Remyelination is one of the most critical yet unmet clinical needs in the treatment of multiple sclerosis.
“While current therapies focus largely on suppressing inflammation, restoring the damaged myelin sheath is essential for preserving neuronal function and preventing long-term disability.”
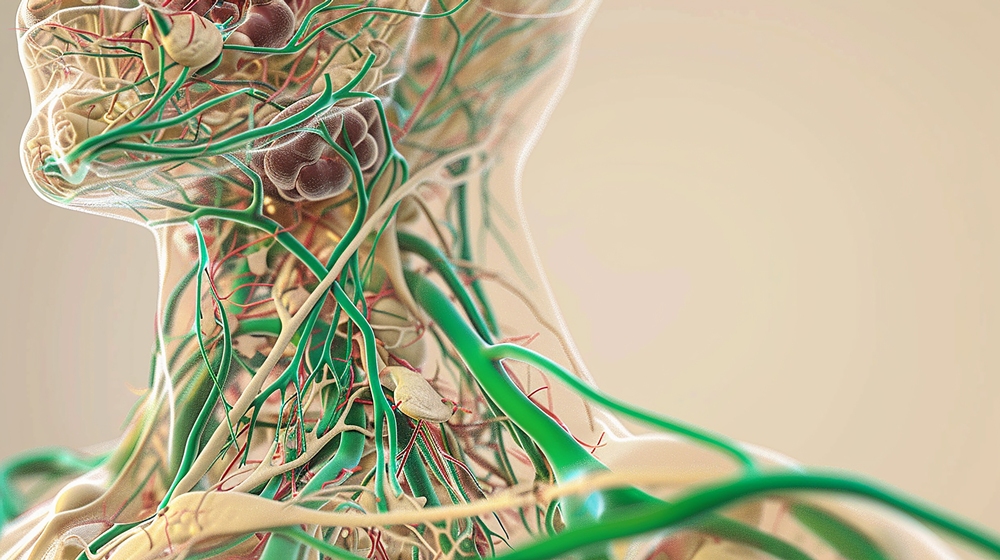
Setpoint’s approach uses electrical stimulation of the vagus nerve, one of the body’s longest nerves, which runs from the head to the abdomen and helps regulate several bodily functions.
The vagus nerve is involved in the inflammatory reflex, a pathway that can help reduce excessive inflammation and restore immune balance.
The device is about the size of an oral capsule and is implanted in the left vagus nerve region of the neck through a small incision. It is powered by a rechargeable battery, which can be charged wirelessly.
Clinicians can adjust the device’s electrical pulses using an iPad application.
Rosenthal said: “Neuroimmune modulation is hypothesised to recalibrate immune activity while supporting the conditions necessary for myelin repair, and offers an innovative avenue to evaluate whether meaningful remyelination can be achieved in patients living with MS.”
In preclinical work involving a rat model of MS-like myelin damage, vagus nerve stimulation was associated with reduced activation of inflammatory cells, including microglia and astrocytes, and improved myelin repair in lesions.
Microglia and astrocytes are support cells in the brain and spinal cord that can contribute to inflammation when overactive.
In the trial, all participants will receive the implanted device. Around two in three will have it activated at the start and will receive stimulation for one minute each day for 48 weeks.
The remaining participants will receive inactive stimulation and serve as a control group. After 48 weeks, they may switch to active stimulation, and all participants will be monitored for a further 48 weeks to assess longer-term safety.
The trial is being conducted under an investigational device exemption from the US Food and Drug Administration. This allows an experimental medical device to be used in a clinical trial to collect safety and effectiveness data.
The FDA has granted the device breakthrough designation for relapsing-remitting MS. The status is intended to speed the development and review of medical technologies for serious or chronically debilitating conditions that may offer advantages over existing options.
The device has also been accepted into the FDA’s Total Product Life Cycle Advisory Program, which gives developers earlier and more frequent contact with regulators.
A version of Setpoint’s system is already approved in the US for certain adults with rheumatoid arthritis.









